From the cellular system harvest to the final Drug Product, several purification steps are implemented in through the Down Stream Process (DSP) to remove HCPs. These residuals proteins are part of the Critical Quality Attribute (CQA) since these residual proteins can affect product quality, stability, efficacy and safety, as well as induce or enhance immunogenicity.
Host cell protein expression is further influenced by culture conditions and methods as well as other aspects of living cells, such as age. The implemented analytical method must be consistent and reliable to perform smaple comparison even if sample matrices are different (different sample complexity between harvest and Drug Substance).
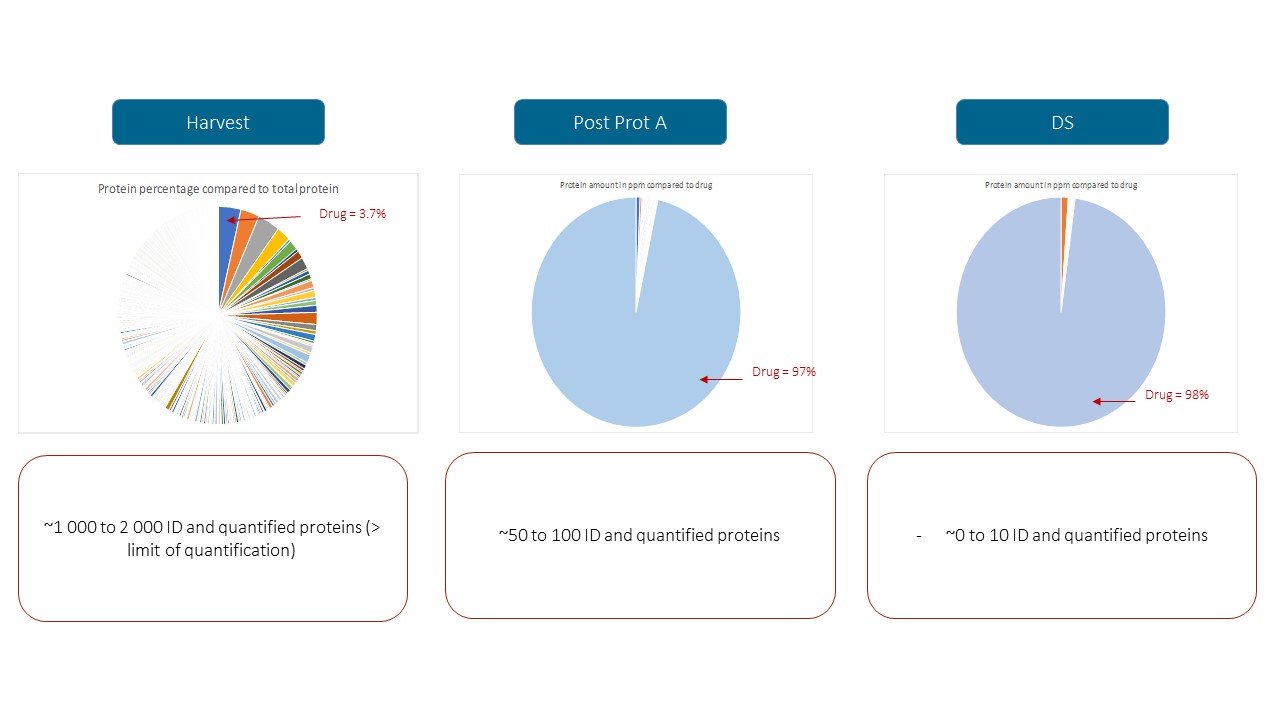
When production processes change, our solution HCP Profiler can be implemented through the whole DSP steps from harvest sample to final drug substance.
It provides for each HCP, an identification and a quantification evaluation allowing to monitor and improve the Downstream Process.
In this example, with HCP Profiler solution we screened all the HCPs at the different purification steps of a therapeutic antibody production. It brought to our customers a better knowledge of its process. The list of individual HCPs and their quantification evaluation give consistent information in order to improve the process. Furthermore, the quality control included in the HCP Profiler solution, ensure reproducibility and accuracy to compare DSP through time.
in this example, the HCP Profiler solution allows to:
- Improve your bioprocess
- Gain knowledge in your HCPs (Individual Identification & Quantification)
- Support your regulatory dossier
 Anaquant HCP analysis I Protein characterisation I Protein analysis
Anaquant HCP analysis I Protein characterisation I Protein analysis