
Introduction
Monoclonal antibodies (mAbs) have emerged as a major class of therapeutics used to treat diseases such as cancer, inflammation or autoimmune diseases. Mass spectrometry is becoming increasingly widespread for the analysis of mAbs, particularly in complex matrices, as it overcomes the cross-reactivity problems associated with conventional techniques based on immunoassays.
Pharma companies conduct pharmacokinetic (PK) at various stages in the development of mAb therapeutics. Anaquant was contracted as part of an early PK study to assess the half-life of a mAb in the plasma of injected rats. Our objective was to specifically and reproducibly determine mAb concentration in plasma samples.
IP-LC-SRM Analytical Strategy Set-Up for mAb quantitation
In order to reach optimal quantitation accuracy and comply with regulatory quantitation limits, we determined the following sample processing procedure:
– Immunoglobulins are purified from plasma samples by immunopurification (IP).
– Immunoglobulins are digested into small peptides, one of which is selected as the signature peptide for specific quantitation of the mAb among all immunoglobulins (classical mass spectrometry bottom-up approach). The sample is subsequently concentrated.
– Then, targeted LC-MS/MS analysis by LC-SRM (Selected Reaction Monitoring) is performed on a triple quadrupole mass spectrometer. The precursor ion corresponding to the entire signature peptide is first selected in the Q1 quadrupole, then fragmented in the Q2 collision cell. Specific fragment ions are selected in the Q3 quadrupole (Figure 1). The precursor ion/fragment ion pair is called a SRM “transition” and provides specificity and sensitivity to the quantitative analysis.
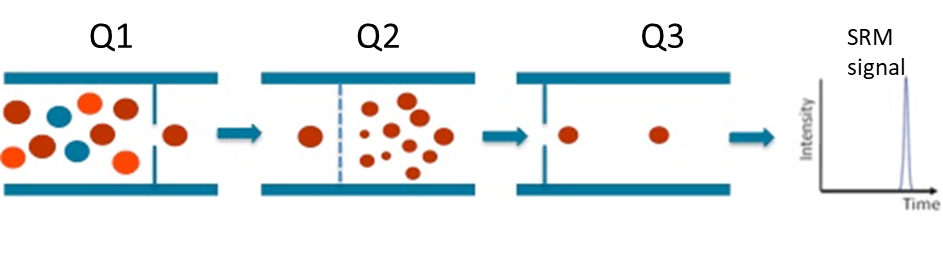
Figure 1 : Schematic diagram of Selected Reaction Monitoring
We set up the quantitation of therapeutic mAb in rat plasma sample using an external range quantification method. A calibration curve (0.5 µg/ml to 100 µg/ml) was therefore established in naive plasma matrix to assess the analytical performances of the developed approach in terms of sensitivity and linear range. Figure 2 validates signal linearity over this concentration range.
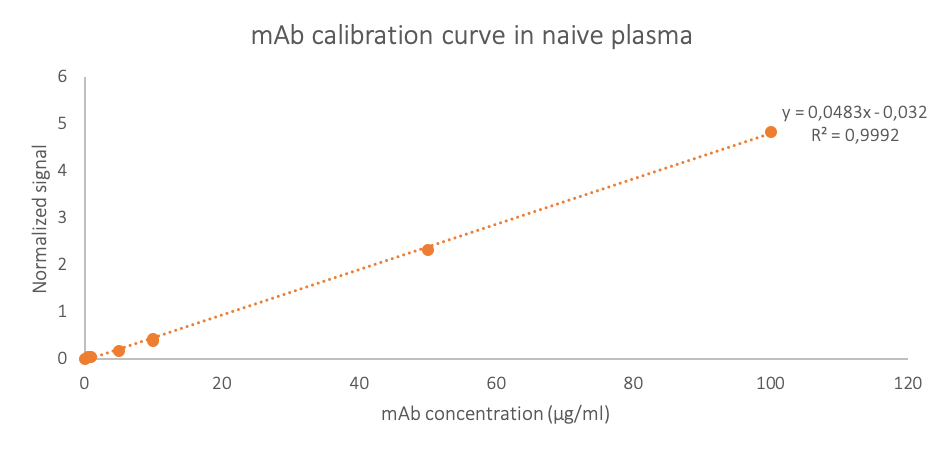
Figure 2 : External calibration curve of therapeutic mAb in naive rat plasma
Results
To assess degradation kinetics, plasma samples were drawn from rats at 10 different time points (from 10 minutes to 28 days) following intravenous administration of therapeutic mAb at 10 mg/kg (n= 2 to 4; 36 samples in total).
Along with the calibration curve points, 50 µl of each of the rat plasma samples underwent our optimized sample processing procedure (IP, enzymatic digestion, peptide concentration prior to LC-MS/MS analysis by LC-SRM).
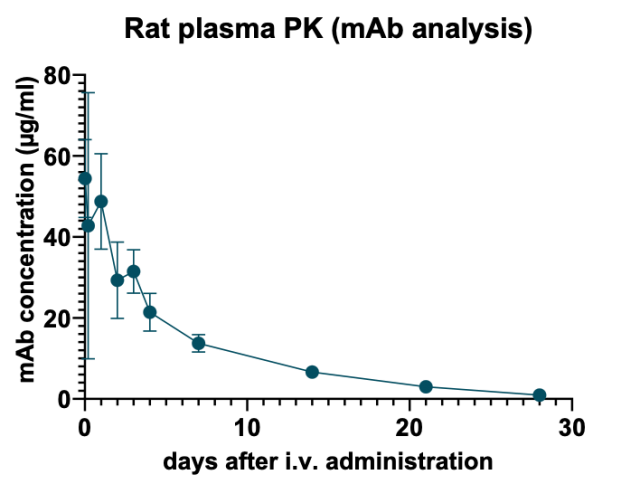
Figure 3 : mAb concentration in rat plasma decreases over time. n= 2 to 4 for each time point
As expected for an intravenously administered antibody, the peak concentration is obtained at the earliest sampling time (10 min). In addition, the plasma drug concentration-time profile shows that plasmatic drug concentration decreases rather homogeneously over time in spite of significant initial inter-animal variability.
These results demonstrate that the degradation kinetics of the therapeutic mAb can be monitored in rat plasma by our IP-LC-SRM method for up to 28 days after drug administration.
Concluding remark
In this study, we demonstrate that our IP-LC-SRM process’s high specificity and sensitivity make it a choice method for early PK study of a therapeutic monoclonal antibody. We have successfully discriminated therapeutic mAbs from endogenous immunoglobulins, and complied with expected quantitation limits.
 Anaquant HCP analysis I Protein characterisation I Protein analysis
Anaquant HCP analysis I Protein characterisation I Protein analysis