
Introduction
Adeno-associated virus (AAVs) is a class of biotherapeutics which use in gene-therapy rapidly increased. As the filed advanced into clinical studies and commercialization, characterization of the AAV drug products, such as host cell proteins (HCPs) identification and quantification, became more and more critical. However, HCP quantification inside AAV samples remains challenging.
ANAQUANT has developed an innovative standard dedicated to MS-based Host Cell Protein profiling analysis: HCPprofiler. This solution is composed by both (1) a hydro-soluble bead coated with 54 peptides based on the READYBEADS technology which, after being spiked directly into the sample, releases peptides at well controlled concentration to obtain a robust and universal calibration curve; and (2) a dedicated software to convert MS signals into amount of each identified protein. Selected peptides are non-natural, their sequences do not correspond to any of the proteome existing in public databases. Those peptides can be spiked in all samples regardless of taxonomy without generating interference.
In this application note we performed HCPs identification and quantification inside HEK293 derived type 2 Adeno-Associated Virus (AAV2) vector thanks to HCPprofiler solution.
Method:
Briefly, protein from capsid and HCPs separation strategy will not be considered because it may generate a risk in HCPs elimination linked to the vector and lead to an underestimated number of HCPs identified. In the implemented strategy, after capsid denaturation, all proteins from samples are enzymatically digested.
Results:
LC-MS analysis of the two different AAV drug products (DP) allowed to identify various number of HCPs inside the two analysed samples: 62 and 30 host cell proteins respectively for DP1 and DP2.
HCPprofiler internal standards added to each Drug Product sample preparation allowed to obtain universal calibration curves used both for sample injection normalisation and individual protein quantification. As presented in Figure 1, two very repeatable calibration curves were obtained in the two AAV samples despite the important matrix differences between the two samples. The added value of the HCPprofiler standard is to be used as quality control for the LC-MS injections: in the two AAV samples both calibration curves pass quality criteria with R² above 0.95 and slope close to 1.00 and sensitivity of the experiment verified by the presence of the low calibration curve points.
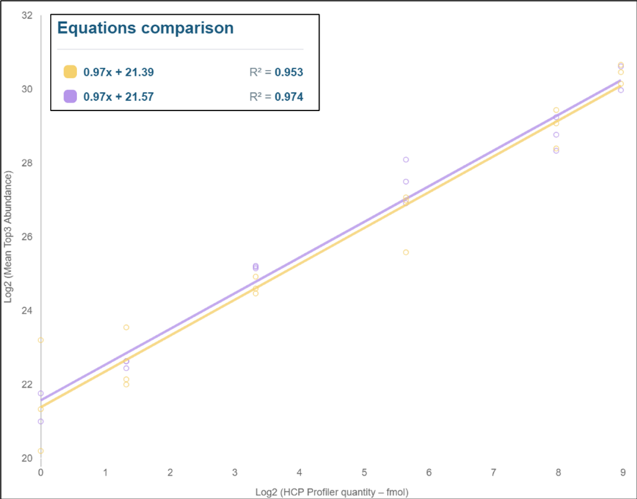
Figure 1 : Calibration curves obtained with HCPprofiler standards in two different AAV drug products.
Individual HCPs quantification was performed allowing to go deeper into the sample characterisation: enabling estimation of HCP abundances in ppm (see Table 1, Figure 2).
| Drug product 1 | Drug product 2 | |
| Number of identified HCP | 62 | 30 |
| Global HCP amount (ppm) | 14539 | 1050 |
| AAV purity (%) | 98.57 | 99.9 |
Table 1 : Global HCP quantification results
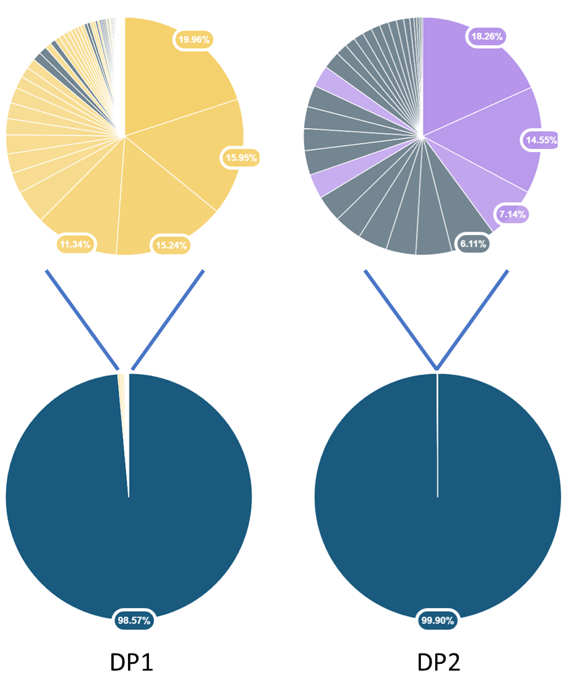
Figure 2 : Pie plots of the AAV purity in each Drug Product sample (down),host cell protein distribution and relative amounts (up).
The overall AAV purity as well as the individual HCP quantities are shown for both AAV samples using the HCPprofiler solution. These results confirmed that the HCPprofiler solution is suitable for characterizing AAV samples regardless of their complexity or drug content. Indeed, DP1 sample contained a much more important quantity of HCPs than DP2, both in terms of number of HCPs and global amount of HCPs (in ppm).
Conclusion:
The analytical workflow presented allows sensitive identification of HCP proteins in AAV2 vector preparations produced in mammalian with Host Cell protein quantification in ppm thanks to HCPprofiler solution. Besides the protein identification capability provided by the use of HCPprofiler solution, the resulting HCPs quantification allowed to go deeper into sample characterization and could highlight potentially immunogenic HCPs contained in HEK293 derived AAV2
The obtained results proved that the HCPprofiler solution is a powerful tool for HCP analyzes in different types of AAV samples and can be an advantage for the optimization of gene therapy product manufacturing.
 Anaquant HCP analysis I Protein characterisation I Protein analysis
Anaquant HCP analysis I Protein characterisation I Protein analysis