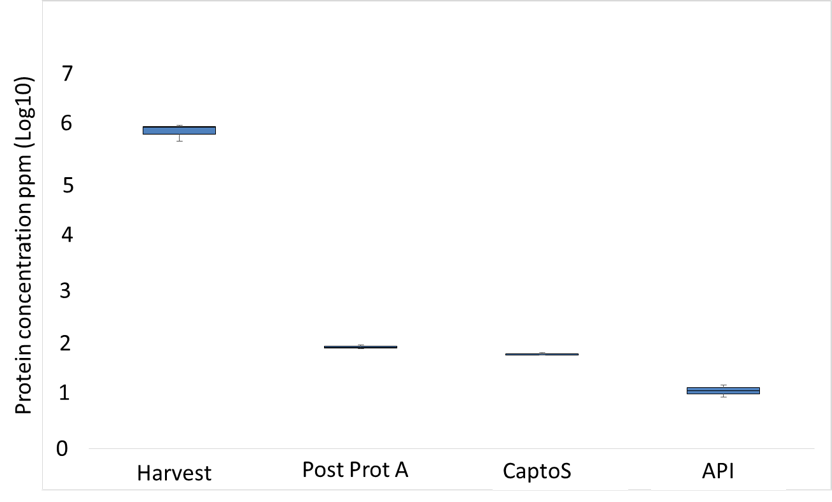
In Biologics production process, from the cellular system harvest to the final Drug Product, several purification steps are implemented through the Down Stream Process (DSP) to remove Host Cell Protein (HCP). These residuals proteins are part of the Critical Quality Attribute (CQA) since these residual proteins can affect product quality, stability, efficacy and safety, as well as induce or enhance immunogenicity. Some specific HCPs, such as lipases, are known to have an impact on final product stability, so it may be relevant to monitor these HCPs in your process.
In this case, we quantified a specific HCP through the different purification steps even if, in this case, the residual protein has a high homology with the product of interest. Our technology permits to detect and asset the HCP to monitor the downstream process.
Since 70 to 80% of biologics are produced in CHO cell lines, we have already developed a method to monitor more than 50 HCPs. This method could be quickly implemented to your process.
Specific HCPs analysis allows you to:
- Support your regulatory dossier
- Improve your bioprocess
- Cross validate Immunoassay
 Anaquant HCP analysis I Protein characterisation I Protein analysis
Anaquant HCP analysis I Protein characterisation I Protein analysis